Desalination

Desalination is the process of desalting water, either by removing the salt from the water or removing the water from the salt. Thermal systems and membrane systems provide about 95% of all desalination globally, with other minor approaches such as freezing and electrodeionization providing the remaining 5%.1Global Water Intelligence, Desalination Markets 2010: Global Forecast and Analysis (Oxford: Global Water Intelligence, 2010). Thermal systems include processes such as boiling water, which separates out the salts by evaporating the water and condensing the subsequent vapor, leaving the salts behind. While effective, this system consumes more energy than other methods. For example, multi-stage flash systems use a series of steps and pressure drops to evaporate water, and multi-effect distillation reuses energy from a previous stage. Thermal approaches have low efficiency in practice, but because they can use waste heat as the source they can be integrated into large facilities with abundant waste heat, such as power plants, improving overall efficiency.

Membrane systems use a semi-permeable barrier to filter out dissolved solids. The most popular membrane approach is reverse osmosis. Other membrane approaches include forward osmosis, nanofiltration, and electrodialysis. Reverse osmosis filters use electrically driven pumps to push water through membranes that act as a filter to separate out the salts. The membranes separate two streams of water with different levels of salinity. Normally, osmotic pressures drive salts from the side with greater salinity toward the side with lower salinity to achieve equilibrium. However, the pumps reverse this flow, causing the salty side to get saltier and the fresh side to get less salty. Reverse osmosis systems are generally more energy-efficient than boiling water, but are expensive technologies that require electricity as opposed to just heat.
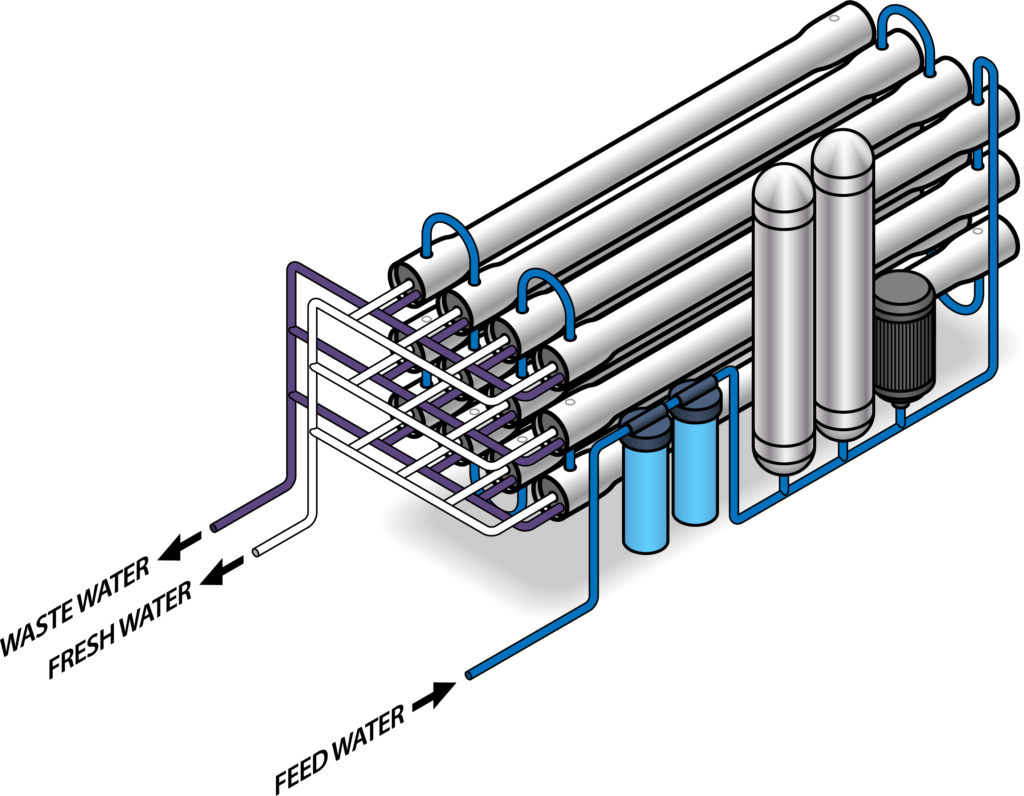
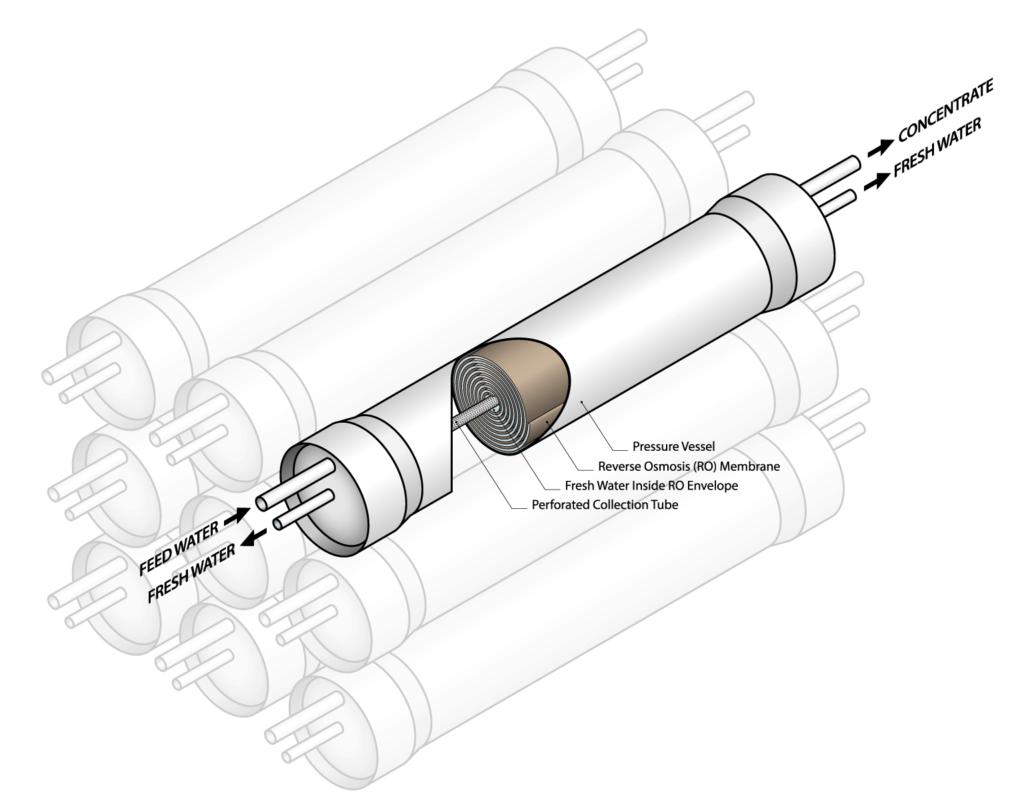
Reverse osmosis systems pump high-pressure saline water through membranes that act as a filter to separate out the salts.
Image Credits: Zern Liew/Shutterstock.com.


